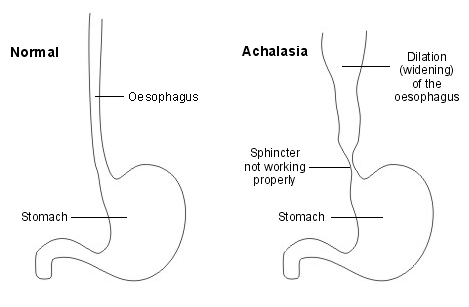
What Is Achalasia
Achalasia
Achalasia is also known as achalasia of cardia, achalasia and cardiospasm, esophageal spasm, esophageal cardiospasm and esophageal achalasia.
The causes of achalasia are multiple. Loss of myenteric nerve fibers in the lower
esophageal sphincter and smooth muscle portion of the esophagus. This has been associated with lymphocytic and eosinophilic infiltrates and fibrosis in later stages of disease.
Loss of intrinsic inhibitory neurons in the myenteric plexus, producing nitric oxide synthase,as well as depletion of networks of interstitial cells of Cajal of the LES, leads to incomplete relaxation.
This motility disorder may be caused by autoimmune degeneration of the esophageal myenteric plexus because association with the HLA class II antigen DQw1 has been noted. Antimyenteric plexus and other antineural antibodies have also been described.
Achalasia is also seen in the rare autosomal recessive disorder Allgrove syndrome (achalasia, alacrima, autonomic disturbance, and acetylcholine insensitivity), which has been linked to a gene mutation on chromosome 12q13. Neurons in this syndrome may be
susceptible to oxidative injury.
Abnormal immune reactions to neurotropic viruses such as varicella zoster, herpes simplex type 1, and measles viruses have been implicated, but the association has not been confirmed. A host T-cell mediated response may lead to neuronal injury.
Symptoms and signs of achalasia are dysphagia (most commonly with both solids and liquids),difficulty belching, regurgitation, chest pain and/or heartburn, globus,frequent hiccups,vomiting of undigested food,symptoms of aspiration such as nocturnal cough; possible dyspnea and pneumonia,weight loss and focal lung examination abnormalities and wheezing may occurs.
The differential diagnosis of achalasia may include Changas disease, vagal injury or surgery, including fundoplication, achalasia-like esophageal dilation has been described after laparoscopic gastric banding , esophageal cancer,infiltrating gastric cancer,oat cell and bronchogenic lung cancer,lymphoma, amyloidosis, paraneoplastic syndrome, angina, bulimia, anorexia nervosa, gastric bezoar, gastritis, peptic ulcer disease
postvagotomy dysmotility, gastroesophageal reflux disease, sarcoidosis, amyloidosis esophageal stricture, esophageal webs and rings, scleroderma, Barrett’s esophagus esophagitis and diffuse esophageal spasm.
Laboratory investigations are vital in confirming the diagnosis of achalasia. Assessment of nutritional status such as omplete blood count, ECG or stress test. Serologic assays for trypanosoma cruzi (Chagas disease) in appropriate individuals.
Imaging technique is considered. Barium swallow with fluoroscopy may demonstrate:
Uncoordinated or absent esophageal contractions or an acutely tapered contrast column (“bird’s beak), dilation of the distal (smooth muscle portion) esophagus and esophageal air fluid level with evidence of poor esophageal emptying. Manometry is generally considered to be the
“gold standard” test to confirm the diagnosis.
High-resolution manometry, or high-resolution esophageal pressure topography, has recently defined subsets of patients with achalasia who may have different responses to medical or surgical therapies. Unlike classic achalasia (Type I), Type II achalasia shows panesophageal pressurization to greater than 30 mm Hg on at least two test swallows, and Type III achalasia shows spastic lumen-obliterating contractions of the distal esophagus on at least two test swallows.
Direct visualization by endoscopy should be performed to exclude other causes of dysphagia, including “functional esophagogastric junction obstruction,” strictures, secondary causes of achalasia, and pseudoachalasia.
Achalasia can be treated. The goals of therapy are to decrease lower esophageal sphincter pressure, relieve symptoms, and prevent progression to a dilated or megaesophagus.
Pneumatic dilation may benefit patients. Multiple sessions may be required, and most protocols use a graded dilation approach, starting with a 30 mm balloon.
Esophageal rupture or perforation is a rare complication that can be managed conservatively in some stable patients.
Surgical: laparoscopic or, now less commonly, open esophagomyotomy is effective Approximately 35% of patients undergoing surgery will develop reflux disease. As a result, some surgeons will perform a “loose” antireflux repair as part of the surgical
procedure. An observational study has suggested that those who have had prior endoscopic treatment before myotomy may not do as well as those who have a primary myotomy.
Early studies suggest that Type I and Type II patients have better treatment responses to these therapies compared with Type III patients.• Endoscopic submucosal myotomy is being explored as another option for treatment.
Medications may be useful for short-term symptom relief and in patients with refractory chest pain. They should only be considered in patients unable to receive, or who are scheduled for, more definitive procedures. lower esophageal sphincter pressure may be lowered bysublingual use of long-acting nitrates or calcium channel blockers.
Achalasia is also known as achalasia of cardia, achalasia and cardiospasm, esophageal spasm, esophageal cardiospasm and esophageal achalasia.
The causes of achalasia are multiple. Loss of myenteric nerve fibers in the lower
esophageal sphincter and smooth muscle portion of the esophagus. This has been associated with lymphocytic and eosinophilic infiltrates and fibrosis in later stages of disease.
Loss of intrinsic inhibitory neurons in the myenteric plexus, producing nitric oxide synthase,as well as depletion of networks of interstitial cells of Cajal of the LES, leads to incomplete relaxation.
This motility disorder may be caused by autoimmune degeneration of the esophageal myenteric plexus because association with the HLA class II antigen DQw1 has been noted. Antimyenteric plexus and other antineural antibodies have also been described.
Achalasia is also seen in the rare autosomal recessive disorder Allgrove syndrome (achalasia, alacrima, autonomic disturbance, and acetylcholine insensitivity), which has been linked to a gene mutation on chromosome 12q13. Neurons in this syndrome may be
susceptible to oxidative injury.
Abnormal immune reactions to neurotropic viruses such as varicella zoster, herpes simplex type 1, and measles viruses have been implicated, but the association has not been confirmed. A host T-cell mediated response may lead to neuronal injury.
Symptoms and signs of achalasia are dysphagia (most commonly with both solids and liquids),difficulty belching, regurgitation, chest pain and/or heartburn, globus,frequent hiccups,vomiting of undigested food,symptoms of aspiration such as nocturnal cough; possible dyspnea and pneumonia,weight loss and focal lung examination abnormalities and wheezing may occurs.
The differential diagnosis of achalasia may include Changas disease, vagal injury or surgery, including fundoplication, achalasia-like esophageal dilation has been described after laparoscopic gastric banding , esophageal cancer,infiltrating gastric cancer,oat cell and bronchogenic lung cancer,lymphoma, amyloidosis, paraneoplastic syndrome, angina, bulimia, anorexia nervosa, gastric bezoar, gastritis, peptic ulcer disease
postvagotomy dysmotility, gastroesophageal reflux disease, sarcoidosis, amyloidosis esophageal stricture, esophageal webs and rings, scleroderma, Barrett’s esophagus esophagitis and diffuse esophageal spasm.
Laboratory investigations are vital in confirming the diagnosis of achalasia. Assessment of nutritional status such as omplete blood count, ECG or stress test. Serologic assays for trypanosoma cruzi (Chagas disease) in appropriate individuals.
Imaging technique is considered. Barium swallow with fluoroscopy may demonstrate:
Uncoordinated or absent esophageal contractions or an acutely tapered contrast column (“bird’s beak), dilation of the distal (smooth muscle portion) esophagus and esophageal air fluid level with evidence of poor esophageal emptying. Manometry is generally considered to be the
“gold standard” test to confirm the diagnosis.
High-resolution manometry, or high-resolution esophageal pressure topography, has recently defined subsets of patients with achalasia who may have different responses to medical or surgical therapies. Unlike classic achalasia (Type I), Type II achalasia shows panesophageal pressurization to greater than 30 mm Hg on at least two test swallows, and Type III achalasia shows spastic lumen-obliterating contractions of the distal esophagus on at least two test swallows.
Direct visualization by endoscopy should be performed to exclude other causes of dysphagia, including “functional esophagogastric junction obstruction,” strictures, secondary causes of achalasia, and pseudoachalasia.
Achalasia can be treated. The goals of therapy are to decrease lower esophageal sphincter pressure, relieve symptoms, and prevent progression to a dilated or megaesophagus.
Pneumatic dilation may benefit patients. Multiple sessions may be required, and most protocols use a graded dilation approach, starting with a 30 mm balloon.
Esophageal rupture or perforation is a rare complication that can be managed conservatively in some stable patients.
Surgical: laparoscopic or, now less commonly, open esophagomyotomy is effective Approximately 35% of patients undergoing surgery will develop reflux disease. As a result, some surgeons will perform a “loose” antireflux repair as part of the surgical
procedure. An observational study has suggested that those who have had prior endoscopic treatment before myotomy may not do as well as those who have a primary myotomy.
Early studies suggest that Type I and Type II patients have better treatment responses to these therapies compared with Type III patients.• Endoscopic submucosal myotomy is being explored as another option for treatment.
Medications may be useful for short-term symptom relief and in patients with refractory chest pain. They should only be considered in patients unable to receive, or who are scheduled for, more definitive procedures. lower esophageal sphincter pressure may be lowered bysublingual use of long-acting nitrates or calcium channel blockers.